|
The power of the modern table lies in its two- or even three-dimensional display of all the known elements (and even the ones yet to be discovered) in a logical system of precisely ordered rows and columns. Yet such lists are simply onedimensional representations. In 1787, for example, French chemist Antoine Lavoisier, working with Antoine Fourcroy, Louis-Bernard Guyton de Morveau and Claude-Louis Berthollet, devised a list of the 33 elements known at the time. Prior to Mendeleev’s discovery, however, other scientists had been actively developing some kind of organizing system to describe the elements. It included 63 known elements arranged according to increasing atomic weight Mendeleev also left spaces for as yet undiscovered elements for which he predicted atomic weights. Yet historians typically consider one event as marking the formal birth of the modern periodic table: on February 17, 1869, a Russian professor of chemistry, Dimitri Ivanovich Mendeleev, completed the first of his numerous periodic charts. The discovery of the periodic system for classifying the elements represents the culmination of a number of scientific developments, rather than a sudden brainstorm on the part of one individual. (In the modern periodic table, a group or family corresponds to one vertical column.) Fortunately, the periodic table allows chemists to function by mastering the properties of a handful of typical elements all the others fall into so-called groups or families with similar chemical properties. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 112 known elements. 
The term “periodic” reflects the fact that the elements show patterns in their chemical properties in certain regular intervals. Remarkably, the periodic table is thus notable both for its historical roots and for its modern relevance. In some instances, new findings initially appeared to call into question the theoretical foundations of the periodic table, but each time scientists eventually managed to incorporate the results while preserving the table’s fundamental structure. But despite the dramatic changes that have taken place in science over the past century-namely, the development of the theories of relativity and quantum mechanics-there has been no revolution in the basic nature of the periodic system. Throughout its long history, the periodic table has been disputed, altered and improved as science has progressed and as new elements have been discovered. The story of the periodic system for classifying the elements can be traced back over 200 years. Indeed, nothing quite like it exists in the other disciplines of science. A version hangs on the wall of nearly every chemical laboratory and lecture hall in the world. The periodic table of the elements is one of the most powerful icons in science: a single document that consolidates much of our knowledge of chemistry. The complete version with artwork is available for purchase here (PDF). To make iodine line up with chlorine and bromine in his table, Mendeleev swapped the positions of iodine and tellurium.Editor's note: The following is a text-only version. However, iodine has similar chemical properties to chlorine and bromine. So iodine should be placed before tellurium in Mendeleev's tables. Iodine has a lower relative atomic mass than tellurium. Its properties were found to be similar to the predicted ones and confirmed Mendeleev's periodic table. 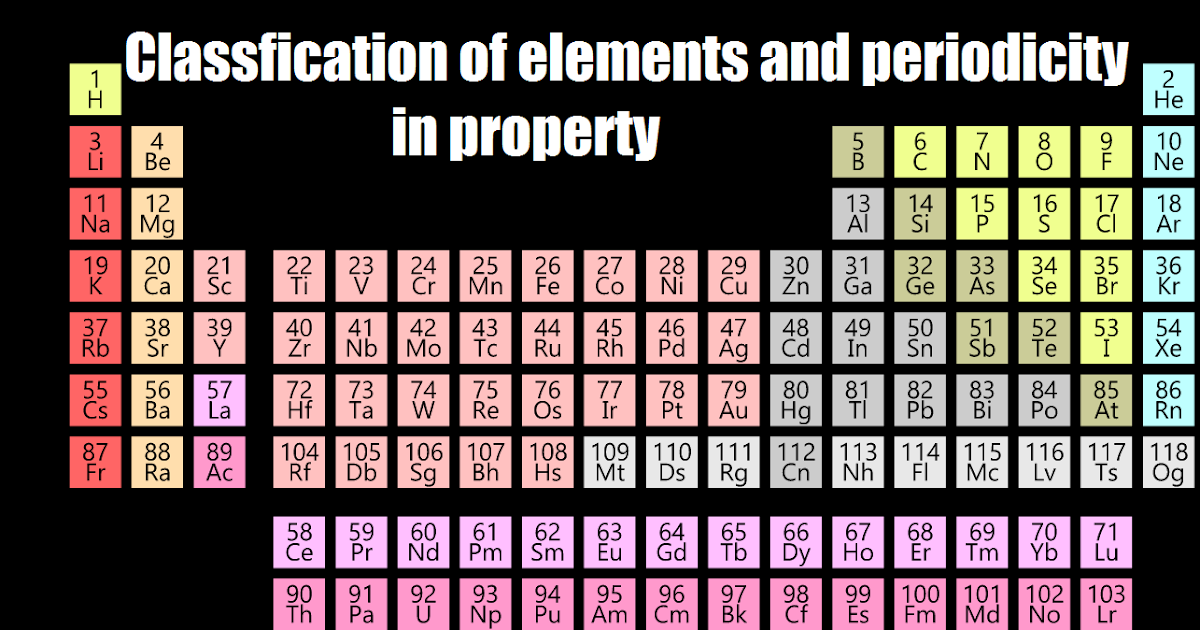
The element germanium was discovered later. For example, Mendeleev predicted the existence of 'eka-silicon', which would fit into a gap next to silicon. By looking at the chemical properties and physical properties of the elements next to a gap, he could also predict the properties of these undiscovered elements. Mendeleev left gaps in his table to place elements not known at the time. Mendeleev's periodic table Predictions using gaps
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
